
This molar mass calculator can only handle two bracket levels at a time. Tricalcium phosphate would be entered as Ca3(PO4)2. The molar mass of MgSO4 (Magnesium sulfate) is: 120.361 grams/mol. It is a white crystalline solid, soluble in water but not in ethanol. For example, calcium carbonate would be entered as CaCO3, not caco3. Magnesium sulfate or magnesium sulphate is a chemical compound, a salt with the formula MgSO4, consisting of magnesium cations Mg2+ (20.19 by mass) and sulfate anions SO2 4. The chemical formula should be entered using standard format. Element: Oxygen Symbol: O Atomic Mass: 15.9994 of Atoms: 1 Mass Percent: 39.696. Element: Magnesium Symbol: Mg Atomic Mass: 24.3050 of Atoms: 1 Mass Percent: 60.304. This calculator is a convenient tool for calculating the molar mass of chemical compounds in lieu of using a periodic table. Molecular weight calculation: 24.3050 + 15.9994. These include consumption of pH adjustment chemicals for RO feedwater, solubilities of scale forming compounds in reverse osmosis systems, and cation rejection calculations using charge balance (meq/l) in nanofiltration systems. Element: Oxygen Symbol: O Atomic Mass: 15.9994 of Atoms: 4 Mass Percent: 53.168. Element: Magnesium Symbol: Mg Atomic Mass: 24.3050 of Atoms: 1 Mass Percent: 20.192. Many other calculations require conversion into moles. Molecular weight calculation: 24.3050 + 32.065 + 15.99944. Knowing the desired concentration of ClO2, the system integrator can calculate the consumption of each of the reactants using the stoichiometric relationship:ĢNaClO2 + NaOCl + 2HCl ↔ 2ClO2 + H2O + 3NaCl Hill system formula: Mg 1 O 4 S 1 CAS registry number: Formula weight: 120.369 Class: sulphate Colour: white Appearance: crystalline solid. For example, certain types of chlorine dioxide (ClO2) generators would use sodium hypochlorite (NaOCl), sodium chlorite (NaClO2) and hydrochloric acid (HCl). Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in CuSO4: Molar Mass (g/mol) Cu (Copper) 1 × 63.546 63.546. Moles of H 2O= (5.15 g/ (246.48 g/mol))(7 mol H 2O/mol MgSO 4 7H 2O) = 0.When calculating consumption of certain RO chemicals for reverse osmosis pretreatment or post-treatment, it is often necessary to convert into moles. Kelsey Ouyang 3H wrote:The formula of Magnesium Sulfate Heptahydrate is MgSO 4 7H 20 (Magnesium ion has a +2 charge, Sulfate has a -2 charge and heptahydrate is 7H 2O since the prefix hepta indicates 7) To prepare 1000 mL of a 0.1 mol/L solution of Magnesium sulfate we have to dissolve 24. 1 The molar mass of Oxygen is 15.999 g/mol. You can see that The molar mass of Aluminum is 26.982 g/mol. Now in MgSO4, there is 1 Magnesium atom, 1 Sulfur atom and 4 Oxygen atoms. Use uppercase for the first character in the element and lowercase for the second character. It will calculate the total mass along with the elemental composition and mass of each element in the compound. Enter the molecular formula of the substance. Then we use Avogadro's constant to find out how many formula units we have of the compound.įormula units = (5.15g/(246.48 g/mol))(6.022x10^23)Ĭ)To determine the moles of water molecules in 5.15 g of Epsom salts, we just need to find out how many moles of the sample there is and multiply it by the mole ratio. So let’s look at the molar mass of Aluminum and Oxygen from the above periodic table. You can see the molar mass value of all the atoms from this periodic table. This program determines the molecular mass of a substance. 
You can see that The molar mass of Magnesium is 24.305 g/mol. So let’s look at the molar mass of Magnesium and Sulfur from the above periodic table. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Now in MgS, there is 1 Magnesium atom and 1 Sulfur atom. Element Magnesium (Mg), Group 2, Atomic Number 12, s-block, Mass 24.305. After finding the amount of moles of the sample, we will then multiply by the mole ratio to find the number of moles of oxygen and then lastly we multiply by Avogadro's constant to find out how many oxygen atoms we haveĪtoms of O=(515 g/ (246.48 g/mol))(11 mol O atoms/1 mol MgSO 4 7H 2O)(6.022x10^23 atoms/mol) = 1.38 x 10^23ī) To find the formula units of the compound we just need to take the amount of sample and divide it by the molar mass to find out how many moles of the sample we have. You can see the molar mass value of all the atoms from this periodic table. To find the atoms of O we need to find how many moles of the magnesium heptahydrate from the sample there is so we divide the amount of grams of the sample by the molar mass. 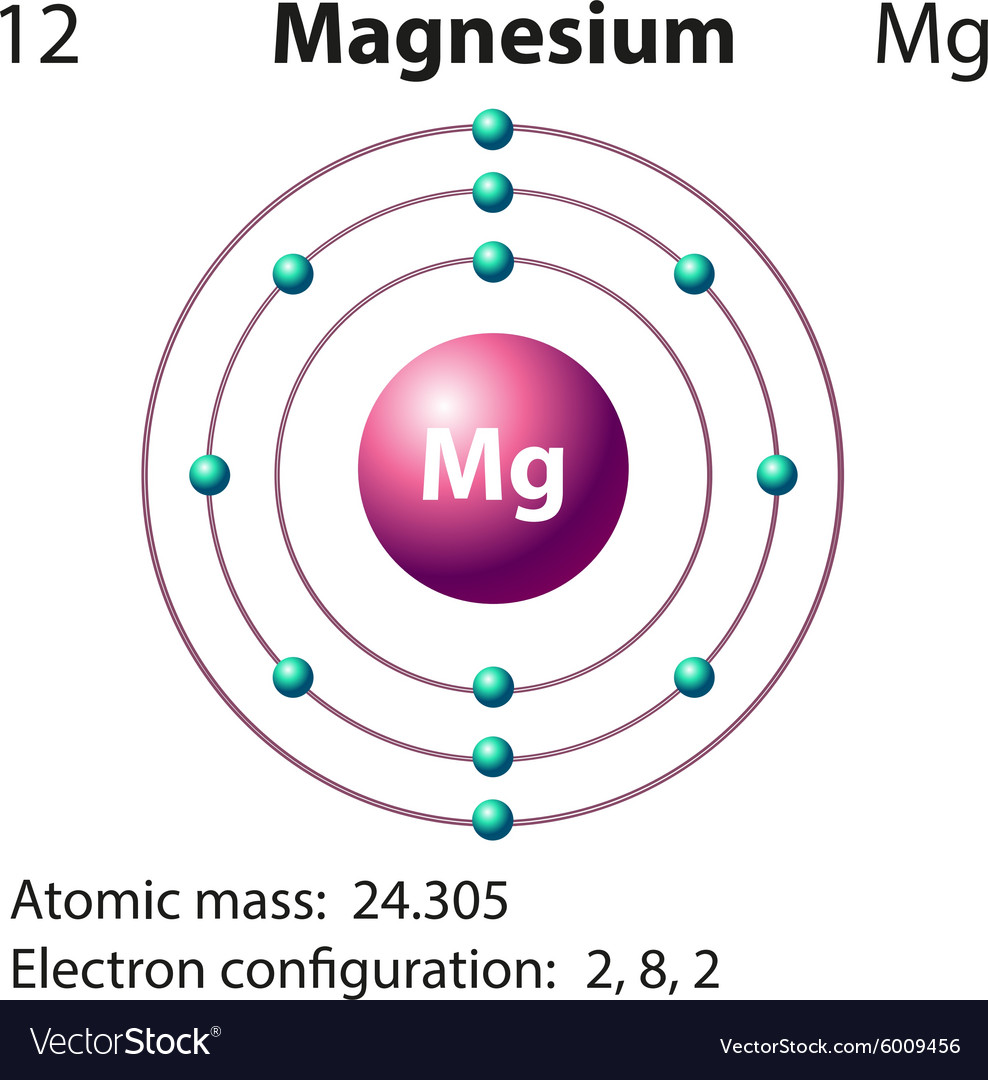 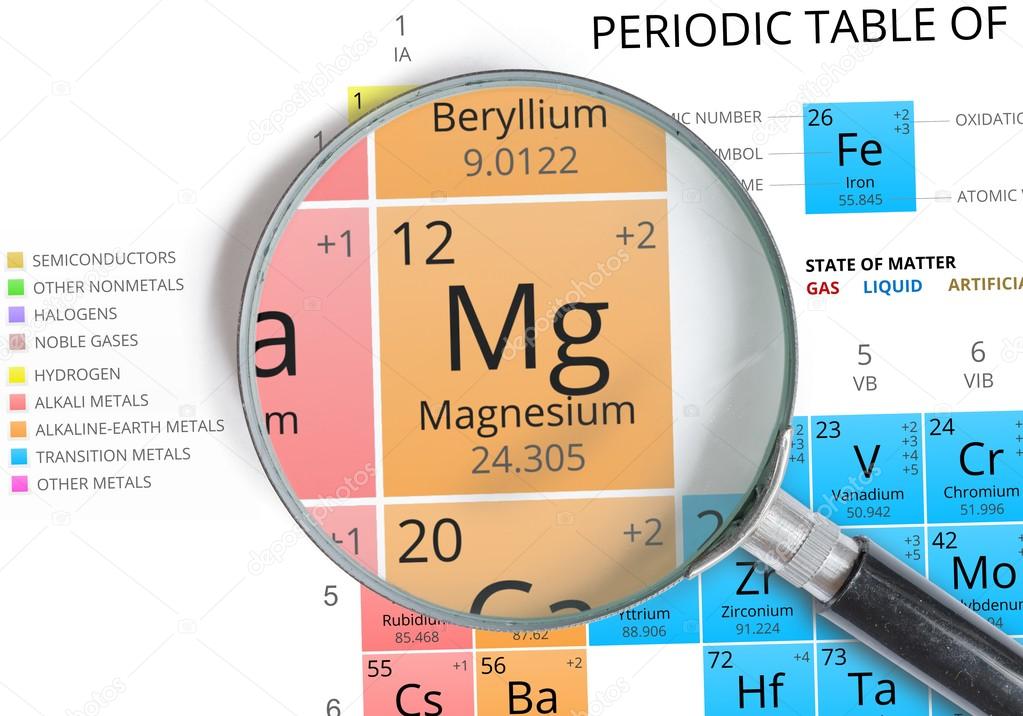
The formula of Magnesium Sulfate Heptahydrate is MgSO 4 7H 20 (Magnesium ion has a +2 charge, Sulfate has a -2 charge and heptahydrate is 7H 2O since the prefix hepta indicates 7)Ī) We first start by finding the formula mass of the magnesium sulfate heptahydrate which is 246.48 g/mol Calculate the molar mass of MgSO4.7H2O in grams per mole or search for a chemical formula or substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
